Last Updated on February 14, 2026
Have you ever wondered how water moves through a membrane or how impurities get filtered out? Understanding the difference between osmosis and reverse osmosis can change the way you see water purification and even impact your health.
This simple yet powerful concept affects everything from the water you drink to how industries clean their water. If you want to make smarter choices about water quality or just satisfy your curiosity, keep reading. By the end, you’ll know exactly how these two processes work and which one matters most for your needs.
Basics Of Osmosis
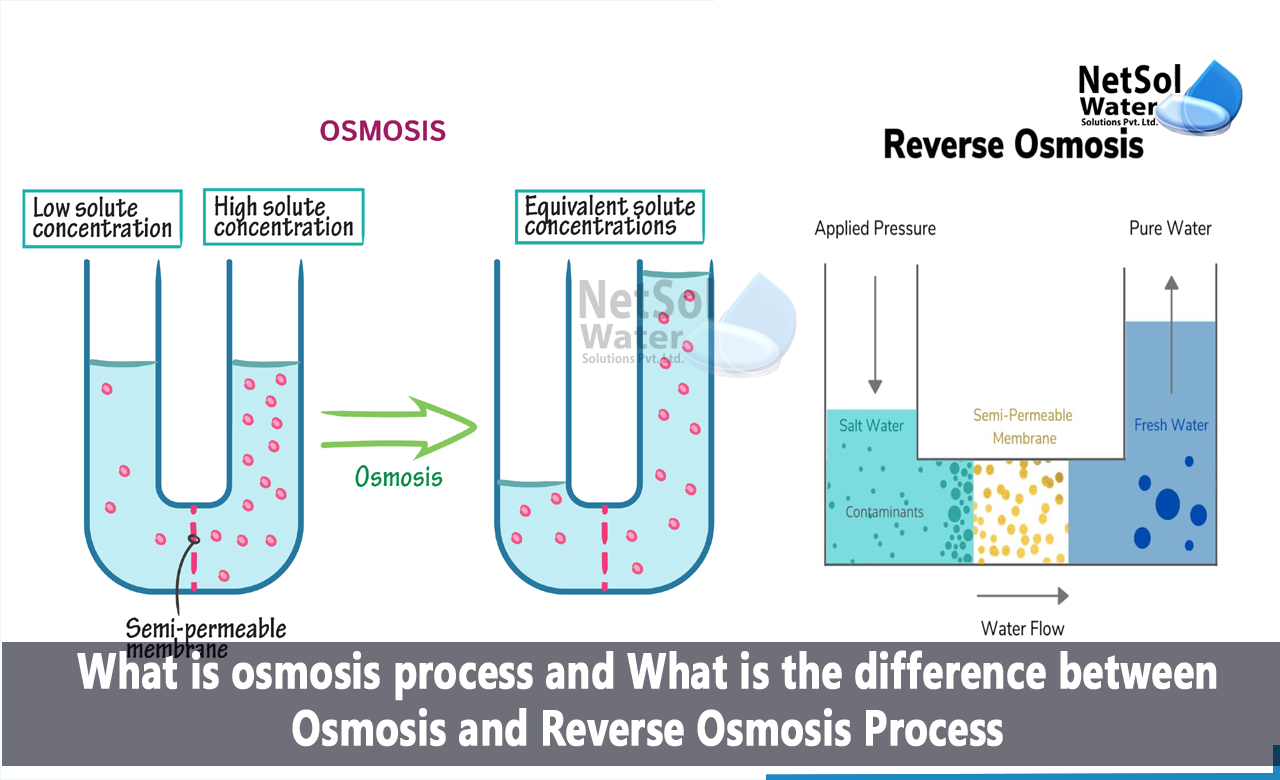
Osmosis is a natural process important to many life forms and systems. It involves the movement of water through a special barrier. This movement helps balance solutions on both sides of the barrier.
Understanding osmosis helps us learn how cells survive and how water purification works. It is the base for technologies like reverse osmosis, which removes impurities from water.
How Osmosis Works
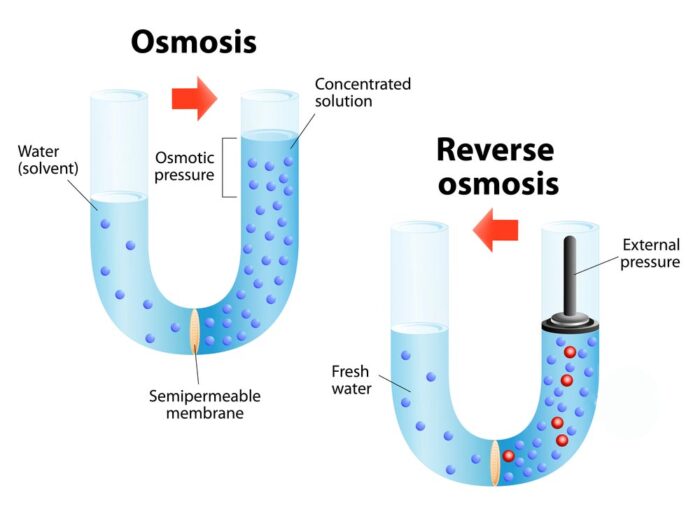
Osmosis happens when water moves from a low concentration of solutes to a high concentration. The water passes through a membrane that allows only water, not solutes. This flow continues until the concentrations on both sides are nearly equal.
Natural Occurrence
Osmosis happens all around us in nature. It occurs in soil, plants, and even in the ocean. Water moves through tiny pores or membranes to balance salt and nutrients levels. This process supports many natural cycles and ecosystems.
Role In Living Organisms
Osmosis is vital for cells in plants and animals. It controls water intake and waste removal. Cells use osmosis to keep their shape and function properly. Without osmosis, cells could dry out or burst.
Basics Of Reverse Osmosis
Reverse osmosis is a popular water purification method. It removes impurities using a special membrane. This process works by applying pressure to force water through the membrane. Only clean water passes, leaving contaminants behind. Understanding how reverse osmosis works helps appreciate its many uses.
Mechanism Behind Reverse Osmosis
Reverse osmosis uses pressure to push water through a semi-permeable membrane. This membrane blocks salts, bacteria, and other impurities. Water molecules are small enough to pass through. The pressure must overcome natural osmotic pressure. This forces water from a concentrated solution to a purer one. The result is clean, safe water ready for use.
Applications In Water Purification
Reverse osmosis is common in home water filters. It removes chlorine, lead, and other harmful chemicals. Many bottled water companies use this method. It improves taste and odor by removing contaminants. It also reduces hardness by filtering calcium and magnesium. This makes water better for drinking and cooking.
Industrial Uses
Industries use reverse osmosis to treat wastewater. It helps recycle water and reduce waste. Power plants use it to purify boiler water. It prevents scale buildup and corrosion. Food and beverage companies use it for ingredient water. It ensures product quality and safety. This method saves costs and protects the environment.
Key Differences In Process
Understanding the key differences in the processes of osmosis and reverse osmosis helps clarify how each method works. Both involve water moving through a membrane, but the forces and directions differ greatly. This section breaks down the main contrasts to give a clear picture.
Direction Of Water Flow
Osmosis moves water from a low to a high concentration area. It flows naturally to balance the concentration on both sides of the membrane. Reverse osmosis forces water in the opposite direction. It pushes water from a high to a low concentration side, against the natural flow.
Pressure Requirements
Osmosis does not need extra pressure. The process depends on natural water movement. Reverse osmosis requires pressure to push water through the membrane. This pressure must be higher than the natural osmotic pressure to work effectively.
Membrane Function
The membrane in osmosis allows only water to pass freely. It blocks many solutes but lets water balance itself. In reverse osmosis, the membrane acts as a strong filter. It blocks most impurities and only lets clean water through under pressure.
Comparison Of Energy Usage
Energy use is a key factor in choosing between osmosis and reverse osmosis. Both processes move water but need different amounts of energy. Understanding these differences helps in picking the right method for water treatment or purification.
Energy costs affect not just the operation but also the environmental impact. Let’s explore how much energy each process uses and why.
Energy Needs In Osmosis
Osmosis is a natural process. It does not require external energy to move water. Water flows from low to high solute concentration by itself. This means osmosis relies on natural pressure differences.
Because of this, osmosis uses very little or no electrical power. It is energy-efficient but slower and less controlled. This process suits situations where energy supply is limited.
Energy Needs In Reverse Osmosis
Reverse osmosis uses pressure to push water through a membrane. This pressure needs energy, usually from electricity. The system uses pumps to create high pressure.
Energy use depends on water quality and system size. Higher pressure means higher energy consumption. Reverse osmosis is energy-intensive but offers faster and cleaner water output.
Impact On Water Quality
Water quality is a key factor in choosing between osmosis and reverse osmosis systems. Both methods affect water differently. Understanding these effects helps pick the right option for clean water.
Effectiveness In Contaminant Removal
Osmosis moves water through a membrane but does not filter out many contaminants. It mainly balances mineral levels. Reverse osmosis uses pressure to push water through a tighter membrane. This removes most impurities like salts, chemicals, and bacteria. Reverse osmosis provides much cleaner water by removing more harmful substances.
Taste And Odor Changes
Osmosis keeps many minerals in water, which can preserve natural taste. It may not reduce bad smells caused by pollutants. Reverse osmosis removes many minerals and impurities, which can make water taste flat or bland. Some users prefer this cleaner taste. Others add minerals back to improve flavor after reverse osmosis treatment.

Credit: www.youtube.com
Common Applications
Osmosis and reverse osmosis serve different roles in daily life and industry. Each process has unique uses based on how they move water and solutes. Understanding their common applications helps to see where each fits best.
Osmosis In Nature And Medicine
Osmosis occurs naturally in plants and animals. Plants absorb water from soil through roots using osmosis. This keeps them hydrated and healthy. In humans, osmosis helps cells maintain balance. It controls water flow in and out of cells. Medical treatments also use osmosis. For example, dialysis machines clean blood by mimicking kidney functions. Osmosis supports life at a basic level.
Reverse Osmosis In Households And Industry
Reverse osmosis filters water by removing impurities. Many homes use reverse osmosis for clean drinking water. It removes salt, dirt, and chemicals. Industries rely on reverse osmosis too. It treats wastewater and produces pure water for factories. Food and beverage companies use it for quality control. Reverse osmosis helps provide safe and pure water everywhere.
Advantages And Limitations
Understanding the advantages and limitations of osmosis and reverse osmosis helps in choosing the right process. Both have unique strengths and weaknesses. These affect their use in water treatment and other fields.
Strengths Of Osmosis
Osmosis is a natural process. It requires no external energy. It helps balance concentration of solutions. Osmosis is simple and cost-effective for basic applications. It supports life in plants and animals by moving water efficiently.
Weaknesses Of Osmosis
Osmosis cannot remove impurities from water. It does not filter out salts or chemicals. The process is slow and depends on natural pressure. It is not suitable for making drinking water safe. Osmosis alone cannot purify contaminated water.
Strengths Of Reverse Osmosis
Reverse osmosis removes most impurities from water. It filters out salts, bacteria, and chemicals. It produces clean and safe drinking water. This process works quickly with applied pressure. Reverse osmosis systems are widely used in homes and industries.
Weaknesses Of Reverse Osmosis
Reverse osmosis requires energy to work. It wastes some water during filtration. The system needs regular maintenance and filter changes. It can remove beneficial minerals from water. Installation and operation costs are higher than osmosis.
Credit: www.netsolwater.com
Credit: www.water-right.com
Frequently Asked Questions
What Is The Main Difference Between Osmosis And Reverse Osmosis?
Osmosis is the natural movement of water through a semi-permeable membrane. Reverse osmosis uses pressure to move water in the opposite direction, filtering out impurities. Both processes involve membranes but serve different purposes in water purification.
How Does Reverse Osmosis Improve Water Quality?
Reverse osmosis removes contaminants like salts, bacteria, and chemicals. It forces water through a fine membrane, leaving impurities behind. This process produces clean, safe drinking water by significantly reducing harmful substances.
Can Osmosis Occur Without External Pressure?
Yes, osmosis happens naturally without any external pressure. Water moves from a low solute concentration to a high solute concentration. This process balances concentrations on both sides of the membrane.
Is Reverse Osmosis Energy-intensive Compared To Osmosis?
Yes, reverse osmosis requires external pressure, often using a pump. Osmosis relies on natural concentration gradients and needs no added energy. Therefore, reverse osmosis consumes more energy than osmosis.
Conclusion
Osmosis and reverse osmosis both move water but in different ways. Osmosis lets water flow naturally to balance things. Reverse osmosis pushes water through a filter to clean it. Each process has its own use and benefits. Understanding these helps you choose the right method.
Clean water matters for health and daily life. Remember, simple science can make a big difference. Choose wisely based on your needs and water quality. Clear water, clear choice.




